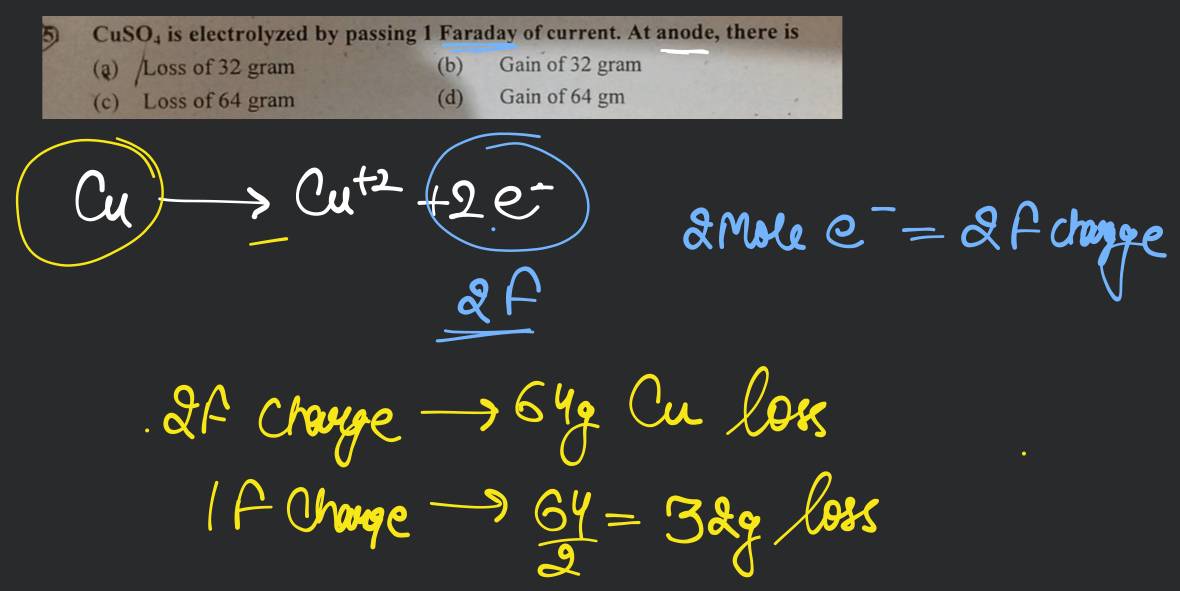The mass of the substance deposited by 1 faraday of electricity is equal to 11 grams. The value of electrochemical equivalent is: A. 11 B. 11 x 96500 - Correct Answers 11 96500 D. data insufficient
the amount of copper deposited by 1 faraday current will be maximum in an acidic solution of 1l of 1.1MCu2Cl2 2.2MCu(NO3)2 3.5M CuSO4 4.5M Cu3(PO4)3 5.10M CuF2

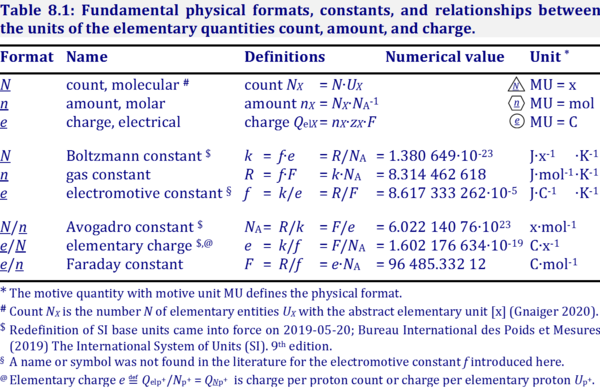
SOLVED: TABLE 13-1: Some Physical Constants and Units Used in Thermodynamics Boltzmann constant, k = 1.381 x 10^(-23) J/K Avogadro's number, N = 6.022 x 10^(23) mol^(-1) Faraday constant, F = 96,480

FARADAY'S LAW. NaCl (s) → Na + (l) + Cl – (l) E° R = V E° O = V 2 Cl - (l) → Cl 2(g) + 2e - 2 Na + (l) + 2e - → 2 Na (s) Electrolytic cell. - ppt download
1.Explain and give e.g. on 'how 1 Faraday will always deposits 1 gm equivalent mass of a substance.'

Cyber Nichel Rame 1 Faraday Tessuto EMF Schermatura 50 "x 3' Materiale di blocco del segnale - Plain Weave : Amazon.it: Casa e cucina

1 mol of charge = 1 Faraday = 96500 coulombs. Represent the cell and the standard emf of the cell having the following reaction. 2Cr(s) +3Ca(aq) = 2Cr + (aq) + 3Cd()

Electrolysis of water with 1 faraday electricity gives | CLASS 12 | ELECTROCHEMISTRY | CHEMISTRY... - YouTube










.png)