
thermodynamics - Why and who has established that $1\, cal \equiv4.186\, J$? - Physics Stack Exchange

Chapter 6. Calorie One calorie expresses the quantity of heat necessary to raise the temperature of 1 g of water by 1° Celsius. Kilocalorie (kCal) - ppt download

SOLVED: The Joule (J) is the SI unit of energy. 1 calorie (cal) = 4.184 J; 1 Calorie (Cal) = 1000 cal = 1 kcal kJ=? J=?





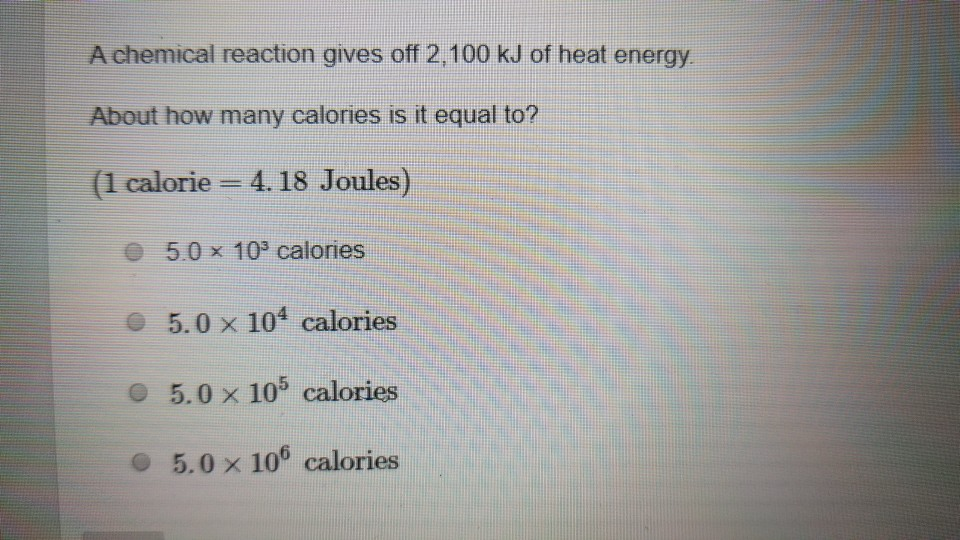
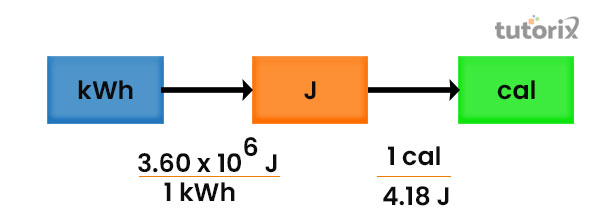




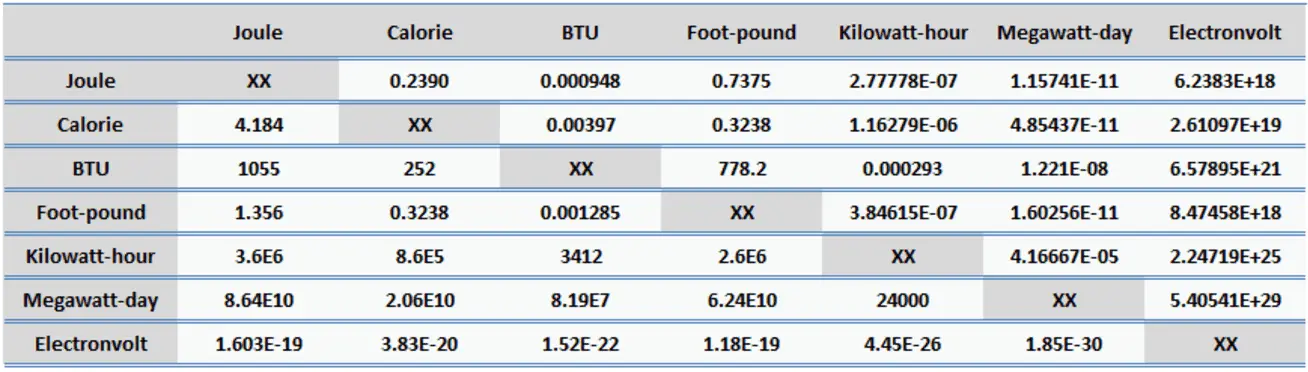


![Bengali] 1 joule is equivalent to calorie. Bengali] 1 joule is equivalent to calorie.](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/16541826.webp)

